XRF Spectroscopy: X-Ray Fluorescence Principles
The Foundation: Atomic Structure and Energy Levels
To understand XRF spectroscopy, we must first grasp basic atomic structure. Atoms consist of a nucleus surrounded by electrons arranged in discrete energy levels or shells designated K, L, M, N, and so forth. The K-shell represents the innermost, lowest energy level closest to the nucleus, where electrons are most tightly bound. Each successive shell (L, M, N) exists at progressively higher energy levels farther from the nucleus with weaker binding forces.
Each element possesses a unique electronic configuration with specific binding energies for electrons in each shell. These binding energies are characteristic and constant for each element—iron's K-shell electrons require precisely 7.11 keV to remove, copper's require 8.98 keV, and lead's require 88.00 keV. This element-specific nature of electron binding energies forms the fundamental basis for XRF's ability to identify elements uniquely.
The electrons in an atom naturally occupy the lowest available energy states, creating stable configurations. When energy is added to the system and an inner-shell electron is removed, the atom enters an unstable, excited state that it immediately seeks to correct through electron transitions from outer shells.
The XRF Process: Excitation and Emission
Primary X-Ray Excitation
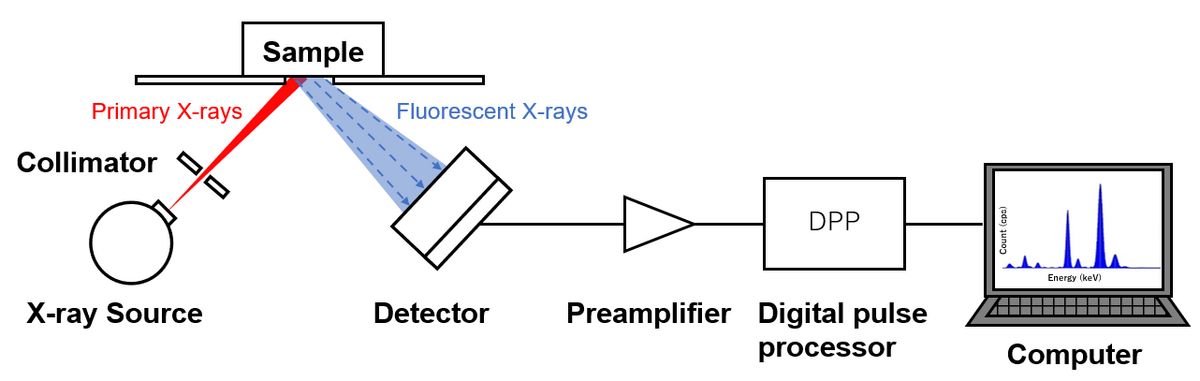
XRF spectroscopy begins when a sample is bombarded with high-energy primary X-rays, typically generated by an X-ray tube. Inside this tube, electrons are accelerated to high velocities and directed toward a metal target (commonly rhodium, tungsten, or silver). When these high-speed electrons strike the target, they produce X-rays through two mechanisms: characteristic X-rays from the target material itself and bremsstrahlung (braking radiation) creating a continuous X-ray spectrum.
These primary X-rays, with energies ranging from a few keV to over 50 keV depending on tube settings, irradiate the sample. When a primary X-ray photon encounters an atom in the sample and possesses energy greater than that atom's electron binding energy, it can transfer sufficient energy to eject an inner-shell electron completely from the atom. This process, called the photoelectric effect, creates a vacancy in the inner electron shell.
The probability of ejecting an electron depends on the primary X-ray energy relative to the electron binding energy. Excitation efficiency peaks when the primary X-ray energy is just above the binding energy of the shell being ionized. If primary X-ray energy falls below the binding energy threshold, ionization cannot occur regardless of X-ray intensity.
Fluorescent X-Ray Emission
Once an inner-shell electron has been ejected, leaving a vacancy in the K-shell (or L-shell, M-shell, etc.), the atom exists in an unstable high-energy state. Nature abhors this instability, and the atom immediately begins to stabilize by having an electron from a higher energy outer shell drop down to fill the inner-shell vacancy.
When an outer electron transitions to fill an inner-shell vacancy, the electron moves from a higher energy state to a lower energy state. The energy difference between these two states must be released, and it emerges as an X-ray photon—the fluorescent X-ray that gives XRF spectroscopy its name. The energy of this fluorescent X-ray precisely equals the difference between the binding energies of the two shells involved in the transition.
Multiple transition pathways are possible. For a K-shell vacancy, an L-shell electron might drop down (producing a Kα X-ray), or an M-shell electron might fill the vacancy (producing a Kβ X-ray). Each transition produces fluorescent X-rays with characteristic energies unique to the element. Iron's Kα fluorescence appears at 6.40 keV, copper's at 8.05 keV, silver's at 22.16 keV—creating elemental fingerprints that enable precise identification.
Detection and Measurement
X-Ray Detectors
The fluorescent X-rays emitted by the sample must be detected and their energies measured to identify and quantify elements present. Modern XRF systems employ sophisticated detectors, with silicon drift detectors (SDD) representing the current state-of-the-art for portable and many benchtop systems.
When a fluorescent X-ray photon enters the detector, it generates electron-hole pairs in the semiconductor material. The number of electrons produced is proportional to the X-ray photon's energy—higher energy X-rays create more electrons. The detector measures this charge and converts it into an electrical signal proportional to the X-ray energy.
Energy-dispersive XRF (EDXRF) systems measure all fluorescent X-ray energies simultaneously using semiconductor detectors, building up a complete energy spectrum over the measurement period. Wavelength-dispersive XRF (WDXRF) systems use crystal diffraction to separate X-rays by wavelength before detection, achieving superior energy resolution at the cost of increased complexity and sequential measurement.
Spectrum Generation
As the detector accumulates fluorescent X-rays from the sample, it builds an energy spectrum—a graph plotting X-ray intensity (number of detected photons) against energy (keV). Elemental peaks appear at characteristic energies corresponding to the fluorescent X-rays emitted by each element in the sample.
The height and area of each peak correlate with the concentration of that element. More atoms of an element in the sample produce more fluorescent X-rays at that element's characteristic energies, creating taller, larger peaks. This relationship between peak intensity and elemental concentration enables quantitative analysis.
Modern detectors achieve energy resolutions around 125-140 eV FWHM (full width at half maximum), allowing clear separation of most elemental peaks. This resolution determines the system's ability to distinguish between elements with closely spaced emission lines and impacts both identification accuracy and quantification precision.
Fundamental Spectroscopy Principles
Characteristic X-Ray Lines
Each element produces a characteristic pattern of X-ray emission lines determined by its electronic structure. Light elements (low atomic number) primarily emit K-series X-rays (Kα, Kβ) resulting from transitions to K-shell vacancies. Heavier elements produce both K-series and L-series X-rays (Lα, Lβ, Lγ), with the L-series arising from transitions filling L-shell vacancies.
For the heaviest elements, M-series lines also become significant. The specific energies of these characteristic lines are tabulated and well-established for all elements, enabling precise identification. When XRF software detects a peak at 6.40 keV, it definitively identifies iron; a peak at 8.64 keV indicates zinc; 22.16 keV signals silver.
Understanding which series of lines to expect from different elements helps analysts interpret spectra correctly. Analyzing geological samples rich in heavy elements like barium, lead, or rare earths requires attention to L-series lines, while metal alloys of lighter elements focus on K-series emissions.
Matrix Effects
XRF spectroscopy faces challenges from matrix effects—situations where the presence of one element affects the measured intensity of another. Absorption effects occur when heavy elements in the sample absorb fluorescent X-rays from lighter elements, reducing their apparent concentrations. For example, iron in a sample strongly absorbs magnesium's fluorescent X-rays, making magnesium appear less concentrated than it truly is.
Enhancement effects (secondary fluorescence) happen when fluorescent X-rays from one element possess sufficient energy to excite additional fluorescence from another element. If a sample contains both iron and chromium, iron's Kα X-rays can excite chromium atoms, producing extra chromium fluorescence beyond that caused by the primary X-ray beam. This makes chromium appear more concentrated.
Modern XRF software incorporates sophisticated algorithms to correct for these matrix effects, either through empirical influence coefficients derived from standards or through fundamental parameter calculations based on atomic physics. Proper matrix correction is essential for accurate quantitative analysis in multi-element samples.
Quantitative Principles
Converting fluorescent X-ray intensities into elemental concentrations requires calibration. The relationship between measured intensity and concentration is not always linear due to matrix effects, making calibration crucial for accurate results.
Empirical calibration uses certified reference materials (standards) with known compositions spanning the concentration range of interest. By measuring these standards, the system establishes mathematical relationships between intensity and concentration for each element. Unknown samples are then analyzed by comparing their measured intensities to these calibration relationships.
The fundamental parameters approach calculates expected fluorescent intensities from first principles using comprehensive databases of atomic properties—fluorescent yields, mass absorption coefficients, transition probabilities. This theoretical method iteratively adjusts assumed sample composition until calculated and measured spectra match, requiring minimal or no calibration standards.
Both approaches have advantages: empirical calibration excels for routine analysis of similar sample types, while fundamental parameters offers flexibility for diverse samples without extensive reference materials. Modern XRF systems often combine both approaches for optimal performance.
Advantages of XRF Spectroscopy
XRF spectroscopy's non-destructive nature preserves samples intact—no dissolution, no cutting, no combustion. Valuable items, finished products, and irreplaceable artifacts undergo analysis without alteration. Speed represents another major advantage, with most analyses completing in seconds to minutes compared to hours for many alternative techniques.
Multi-element capability allows simultaneous detection and quantification of dozens of elements from a single measurement. This comprehensive analysis efficiency saves time and resources compared to sequential single-element methods. The technique requires minimal sample preparation—often just a clean, flat surface—reducing complexity and labor.
Portability has advanced dramatically with modern handheld XRF analyzers bringing laboratory-quality analysis to field locations, production floors, and remote sites. This mobility enables real-time decision-making exactly where materials exist, transforming quality control and verification workflows across industries.
Limitations and Complementary Techniques
XRF spectroscopy struggles with light elements below sodium (atomic number 11) because their weak fluorescent X-rays are absorbed by air and detector windows. Helium purging or vacuum systems extend capabilities toward lighter elements, but carbon, nitrogen, and oxygen remain challenging for standard XRF.
Surface analysis depth is limited to approximately 10-100 microns depending on sample density and elemental composition. XRF measures surface and near-surface regions, not bulk composition throughout thick samples. Samples must be homogeneous within the analyzed volume or require multiple measurement points.
For ultimate trace element sensitivity below parts per million, or for specialized applications requiring different information, complementary techniques like ICP-MS (mass spectrometry), ICP-OES (optical emission), or combustion analyzers extend analytical capabilities beyond XRF's range.
Conclusion
XRF spectroscopy's fundamental principles—photoelectric effect causing inner-shell electron ejection, followed by electron transitions generating characteristic fluorescent X-rays unique to each element—create a powerful analytical technique applicable across countless fields. Understanding these principles illuminates why XRF achieves rapid, accurate, non-destructive elemental analysis from magnesium through uranium.
The physics governing X-ray interaction with matter, combined with sophisticated detection technology and advanced data processing algorithms, transforms XRF from a theoretical curiosity into an indispensable practical tool. Whether analyzing metal alloys in manufacturing, precious metals in jewelry stores, contaminants in environmental samples, or minerals in geological exploration, XRF spectroscopy delivers reliable results based on unchanging atomic physics.
As detector technology continues advancing and software algorithms grow more sophisticated, XRF spectroscopy's capabilities expand while becoming more accessible to users. The fundamental principles remain constant, but their application grows ever more powerful, ensuring XRF spectroscopy's position as a cornerstone technique in modern analytical science for decades to come.




